The spectrum of elastomers range from very simple forms, like the natural rubber already in use in the 19th century, to modern, high performance elastomers from the second half of the 20th century. They are continually being improved.
This blog article will be the first in a two-part series discussing the many different elastomer materials available today, as discussed in Freudenberg's The World of Freudenberg Sealing Technologies.
Natural Rubber (NR)
 In its original form, latex is greasy and sticky. Natural rubber is an elastic polymer, built on isoprene as its monomer. To make it usable technically, the long polyisoprene chains of natural rubber are cross-linked with one another through vulcanization – which involves the addition of sulfur under pressure and heat.
In its original form, latex is greasy and sticky. Natural rubber is an elastic polymer, built on isoprene as its monomer. To make it usable technically, the long polyisoprene chains of natural rubber are cross-linked with one another through vulcanization – which involves the addition of sulfur under pressure and heat.
In the process, sulfur atoms insert themselves between the double bonds of adjacent chain molecules – the majority of the double bonds are maintained. The result is a three-dimensional molecule network whose parts can only shift against one another within certain limits.
The material’s properties can be controlled by changing the number of sulfur atoms within a bridge. Highly dynamic components generally have longer sulfur bridges, while setting behavior and heat stability improves with shorter sulfur bridges. Natural rubber is the rubber with the longest molecule chains, which gives it very good dynamic qualities.
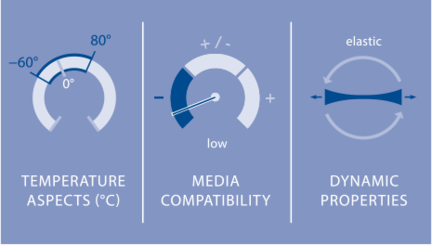 Elastomers made of pure natural rubber have extreme stretchability, ranging from 700 to 1000 percent. At the same time, an elastomer is nearly non-compressible – it does not allow itself to be compressed – only displaced. Natural rubber is stable in acids, bases, water, alcohol and air, that is, in polar media. It has low media compatibility with nonpolar media such as petroleum-based oils, synthetic lubricants, brake fluids based on glycol, overheated moisture, and aromatic hydrocarbons (gasoline). Natural rubber exhibits high elasticity as well as good cold and damping behavior in a temperature range from −60 °C to 80 °C. Natural rubber displays little resistance to high temperatures in combination with oxygen. The reactive double bonds still remaining after vulcanization react easily with oxygen and break down the natural rubber.
Elastomers made of pure natural rubber have extreme stretchability, ranging from 700 to 1000 percent. At the same time, an elastomer is nearly non-compressible – it does not allow itself to be compressed – only displaced. Natural rubber is stable in acids, bases, water, alcohol and air, that is, in polar media. It has low media compatibility with nonpolar media such as petroleum-based oils, synthetic lubricants, brake fluids based on glycol, overheated moisture, and aromatic hydrocarbons (gasoline). Natural rubber exhibits high elasticity as well as good cold and damping behavior in a temperature range from −60 °C to 80 °C. Natural rubber displays little resistance to high temperatures in combination with oxygen. The reactive double bonds still remaining after vulcanization react easily with oxygen and break down the natural rubber.
Natural rubber is mainly used in vibration control technology as a torsion and vibration damper, in engine mounts, machine bearings, diaphragms and damping elements.
Nitrile Rubber (NBR)
 NBR is the abbreviation for nitrile butadiene rubber. This is a synthetic rubber that was developed during the 1930s. It is fabricated through the co-polymerization of acrylonitrile (ACN) (polar building block) and 1.3-butadine (nonpolar building block). Its attributes mainly depend on its ACN content. The manufacturer can vary the content and thus produce different types of the material. In the process, the ACN content determines the balance between media resistance and low-temperature flexibility. Low ACN content produces a low glass transition temperature but also leads to greater swelling in oils and fats. As the ACN content increases, this behavior reverses direction – due to the polar components in the polymer.
NBR is the abbreviation for nitrile butadiene rubber. This is a synthetic rubber that was developed during the 1930s. It is fabricated through the co-polymerization of acrylonitrile (ACN) (polar building block) and 1.3-butadine (nonpolar building block). Its attributes mainly depend on its ACN content. The manufacturer can vary the content and thus produce different types of the material. In the process, the ACN content determines the balance between media resistance and low-temperature flexibility. Low ACN content produces a low glass transition temperature but also leads to greater swelling in oils and fats. As the ACN content increases, this behavior reverses direction – due to the polar components in the polymer.
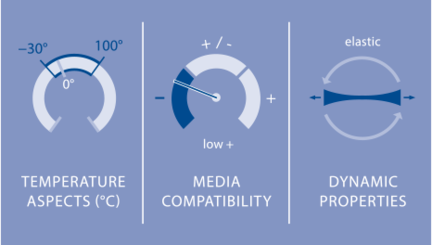 NBR is stable in petroleum oils and greases, in vegetable and animal oils and fats, in hydraulic fluids (HFA¹, HFB² and HFC³), in aliphatic hydrocarbons (propane, butane, gasoline, diesel fuel). NBR displays low media resistance in polar media such as solvents (acetone, MEK, ethyl acetate, diethyl ether), glycol-based brake fluid, superheated steam, HFD pressure fluids, ultraviolet light, and ozone.
NBR is stable in petroleum oils and greases, in vegetable and animal oils and fats, in hydraulic fluids (HFA¹, HFB² and HFC³), in aliphatic hydrocarbons (propane, butane, gasoline, diesel fuel). NBR displays low media resistance in polar media such as solvents (acetone, MEK, ethyl acetate, diethyl ether), glycol-based brake fluid, superheated steam, HFD pressure fluids, ultraviolet light, and ozone.
Nitrile rubber exhibits superior mechanical strength with high elasticity as well as good deformation behavior in a temperature range between −30 °C and 100 °C. Its relatively low heat resistance can be traced to the presence of reactive double bonds. It is used for oil- and fuel-resistant seals such as Simmerrings®, O-rings and frame seals. The use of NBR marked the arrival of elastomers as a sealing material particularly in general industry. It is also employed in the auto industry.
Hydrogenated Nitrile Rubber (HNBR)
 HNBR is produced through the selective hydrogenation¹´² of NBR. The reactive double bonds are removed from NBR. As a result, the molecule chain does not react as easily to oxygen and is thus significantly more heat resistant than NBR. In addition, HNBR offers better wear behavior in dynamic applications.
HNBR is produced through the selective hydrogenation¹´² of NBR. The reactive double bonds are removed from NBR. As a result, the molecule chain does not react as easily to oxygen and is thus significantly more heat resistant than NBR. In addition, HNBR offers better wear behavior in dynamic applications.
For example, HNBR is also resistant to hot water and exhibits good cold and high temperature behavior between –40 °C and 140 °C. In sealing technology, 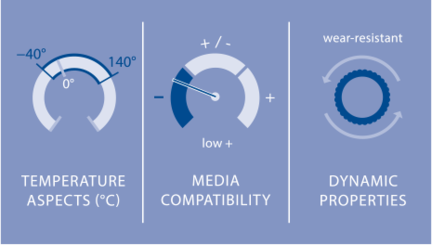 the significantly more expensive HNBR, which is a proven standard material for automotive engine timing belts, is primarily used in applications where higher temperatures and/or mechanical strength are required. Its strength is above all in demand for rod and piston seals, along with O-rings, radial shaft seal rings, boots and diaphragms. Especially in the processing industry, HNBR offers wide-ranging opportunities for use.
the significantly more expensive HNBR, which is a proven standard material for automotive engine timing belts, is primarily used in applications where higher temperatures and/or mechanical strength are required. Its strength is above all in demand for rod and piston seals, along with O-rings, radial shaft seal rings, boots and diaphragms. Especially in the processing industry, HNBR offers wide-ranging opportunities for use.
If you have any questions regarding seal elastomers, please feel free to contact us, and our engineering department will follow up with you.
